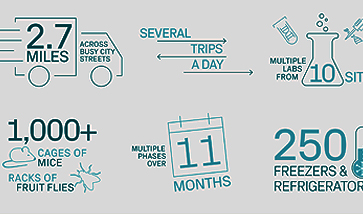
As cell and gene therapies (CGT) advance into the clinical stage, many cold chain complexities arise not only for these novel products but also for the biological samples that must be collected from patients during clinical trials. Managing these sensitive materials—including storage, transportation, and tracking—requires robust and adaptable systems to ensure safety, integrity, and regulatory compliance that are essential for FDA review.
We interviewed Kathi Shea, Senior Director of Repository Innovation at Azenta Life Sciences, to learn how biotech companies can better navigate the clinical phases of CGT development.
What logistical challenges do clinical trials bring to CGT development?
Clinical trial sponsors need to coordinate the delivery of investigational medicinal products to clinical sites and the collection of samples from participants after they receive treatment. These processes are intertwined, and it’s an advantage to have a partner who can support activities from Phase 0 through Phase 3 and beyond.
When storing and handling clinical-phase samples and materials, their fragility cannot be overstated. Retrieving materials manually from a liquid nitrogen freezer can quickly warm the targeted and non-targeted samples in unintended ways. Repeated exposure over time has a compounding effect as well. If I move a box of samples from -180˚C to ambient conditions, I basically have 90 seconds before it starts crossing the glass transition temperature, the point at which degradation occurs. Automated units, on the other hand, can keep biological materials well below that critical temperature throughout the entire retrieval process, significantly lowering the risk of degradation.
How do you maintain the quality of advanced therapies, many of which have a very short shelf life and demanding requirements for storage?
You need a strong quality management system that meets all regulatory requirements, including Good Manufacturing Practices (GMP) and Good Tissue Practices (GTP) for those considered blood products. Also, full traceability from the moment a material is shipped all the way through to end distribution is crucial. Finally, a network of global facilities with automation and highly trained personnel provides a CGT company with the fastest path to advance their programs.
What kinds of safeguards are needed for storing and managing CGT materials and clinical samples?
An inventory management system should be set up with various safeguards and risk mitigation protocols. If you have defined that a project should have a group of materials at a certain temperature, the system shouldn’t allow you to store it at a different temperature.
From an infrastructure standpoint, you need to do everything that adds to the security and stability of the temperature within the storage units. For example, liquid nitrogen freezers, whether automated or manual, should be fed by vacuum-insulated piping, which ensures immediate delivery of LN2 upon demand. All lines should stay primed all the time so that cold LN2 is always reaching the freezers. Having large LN2 tanks to source the units in the event of any disruption to liquid nitrogen deliveries adds another layer of protection.
What about regulatory compliance for advanced therapies?
Cell and gene therapies are viewed as innovative therapies by the FDA. For regulatory purposes, they may be treated more like a pharmaceutical or more like a blood product. A quality system that is set up to handle both is required. There is also a strong need to obtain state licensure to support distribution globally for both types of products. Customers also benefit from additional accreditation and certifications like ISO, CAP, EMA, and PMDA.
Azenta Life Sciences provides a comprehensive range of solutions across every phase of CGT development. Learn how we can support the rigor of your program all the way from target discovery through scale-out.
About the Guest
Kathi Shea

Kathi Shea is the Senior Director of Repository Innovation at Azenta Life Sciences. She has served on the ISBER Board for 8 years in various roles, including Director, Secretary Treasurer, and President. She has 30 years of experience leading biorepository programs and advising organizations on the design of biorepositories and quality systems as well as optimal methods for collection, preservation, and annotation of biospecimen collections.