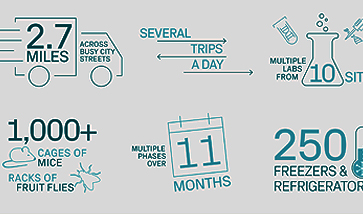
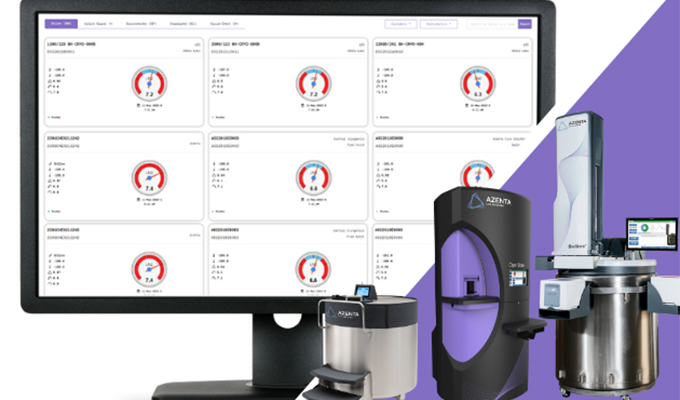
Building Cell Therapy at Scale: Saudi Arabia’s Vision for a Sustainable Advanced Therapies Ecosystem
Part 29 of our Bridging the Gap Series (Summary of the webinar session held in April 2026) For all the scientific progress in cell and gene therapy, one of the field’s biggest challenges remains the same: how to turn innovation into sustainable patient access. That question is especially important in regions where healthcare systems are growing quickly, […]
View Blog