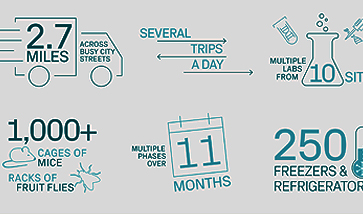
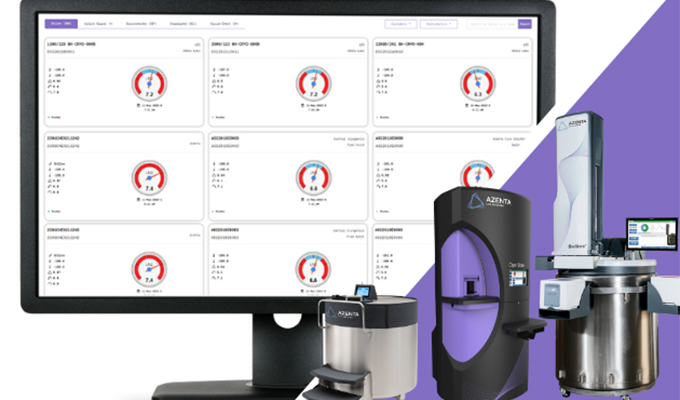
Foundational Strategies That Can Shave Months Off Your Clinical Program: Biorepositories
Part 2 of our Strategies to Speed Clinical Trials Series The path from therapeutic discovery to regulatory approval remains long, with significant time devoted to clinical testing. When investing up to 15 years or more in a potential therapeutic, with much of that time dedicated to clinical trials1, any opportunity to save time during clinical phases can […]
View Blog