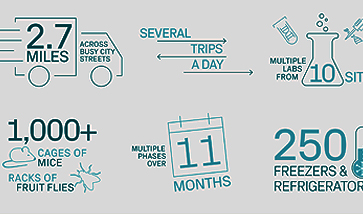
Part 1 of our Strategies to Speed Clinical Trials Series
In the ever-competitive drug development landscape, time is a precious and limited resource. During often-complex clinical trials, every delay can have a real-world impact on patients, healthcare systems, and the bottom line. In fact, one recent review determined that reducing clinical development time from 10 months to 6 months had a value of approximately $100 million on the open market1.
A very effective way to reduce clinical trial delays is through a close partnership between stakeholders and biorepositories where each entity applies their respective strengths to the clinical project. With clearly defined and executed responsibilities from each side, clinical projects can benefit from streamlined operations, reduced redundancies, and increased time savings. This is possible while maintaining high levels of patient safety and regulatory compliance.
In this first of our two-part blog series, we discuss two strategies that clinical contributors like biopharma and cohort organizations can implement to create time-saving opportunities before the first clinical sample is even collected.
Two Biopharma Strategies: Early Clinical Study Decisions Drive Time-Saving Advantages
The foundation for efficiency and time-savings can be laid long before the first patient is enrolled or the first clinical sample is collected. Early decisions around study design, patient consent frameworks, and long-term sample utilization can help to maximize clinical specimen value over time and bypass downstream operational pain points.
Biopharma Strategy #1: Build Future Study Flexibility into Patient Consent
Informed consent is a legal, regulatory, and ethical obligation in clinical studies. And although it is a vital element of any study, obtaining and documenting patient consent can take considerable time. Even before gathering informed consent, simply recruiting patients can be difficult and time-consuming2.
Instead, biopharma companies can build meaningful flexibility by working with their regulatory and ethics teams to draft broader yet transparent and patient-centric consent language that permits patient specimens to be repurposed for projects such as:
- Future potential use
- Secondary analyses
- Cross-study utilization
This flexibility shouldn’t be misconstrued as blanket permission for undefined uses, but rather it is a means to anticipate and prepare for scientific and commercial decisions that inevitably emerge later.
When stored at a qualified biorepository, the resulting sample library remains usable beyond the scope of a single clinical study and may be scaled at any time. Without the need for additional patient recruitment or follow-on informed consent efforts, potentially significant time savings may be reaped during the initial clinical trial and other or subsequent studies.
When stored at a qualified biorepository, the resulting sample library remains usable beyond the scope of a single clinical study and may be scaled at any time.
Biopharma Strategy #2: Plan Beyond Immediate Clinical Sample Needs
With flexible patient consent systems in place, biological specimens are no longer limited to single-use assets. This unlocks enormous time saving efficiencies like:
- Reducing recruitment efforts and timelines as fewer new patients are needed
- Eliminating sample recollection and reprocessing steps in the lab
- Accelerating data generation with near immediate material availability
In future studies, a sample library may be filtered for a desired biomarker to save time and effort instead of recruiting, testing, and screening new patients. Sample reuse for future non-interventional studies is only as powerful as the team’s ability to coordinate with other stakeholders and anticipate potential sample use beyond the active study. This forethought also helps to inform sample information like total volume needed, allocation, storage conditions, and aliquoting protocols.
By treating biospecimens as long-term assets that may be used multiple times, it may be possible to reduce clinical studies by weeks or even months. In a competitive race, this time savings could profoundly impact commercialization success.
By treating biospecimens as long-term assets that may be used multiple times, it may be possible to reduce clinical studies by weeks or even months.
More Time-Saving Strategies
From building flexibility into patient consent to enabling sample reuse across multiple studies, these proactive strategies can help transform samples from single-use resources to long-term assets. The result is smarter use of clinical study time, reduced delays and redundancies, and a more efficient path to clinical and commercial success.
In the second installment of this blog series, we’ll explore how partnering with a biorepository can play a critical role in further eliminating delays and time-consuming clinical bottlenecks through centralized management, standardized processes, and streamlined logistics.
Explore more in the Strategies to Speed Clinical Trials Series
References
- Brown DG, Wobst HJ, Kapoor A, Kena LA, Southall N. Clinical development times for innovative drugs. Nat Rev Drug Discov. 2022 Nov;21(11):793-794. doi: https://doi.org/10.1038/d41573-021-00190-9
- Hung M, Mohajeri A, Almpani K, Carberry G, Wisniewski JF, Janes K, Janes B, Hardy C, Zakeri G, Raymond B, Trinh H, Bretner J, Cheever VJ, Garibyan R, Bachstein P, Licari FW. Successes and Challenges in Clinical Trial Recruitment: The Experience of a New Study Team. Med Sci (Basel). 2024 Aug 14;12(3):39. doi: https://doi.org/10.3390/medsci12030039