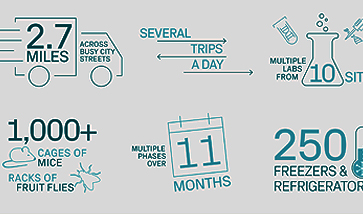
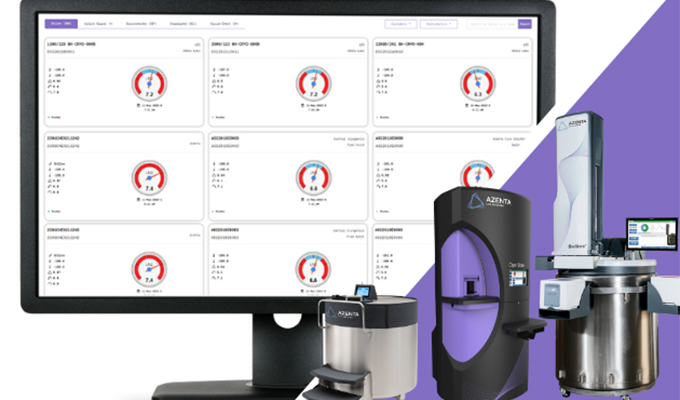
Setting the Global Standard for Patient Care in Cellular Therapies
Part 2 of our Bridging the Gap Series (Summary of the webinar session held in December 2023) As the cell therapy industry continues to expand, one organization is leading the way in advanced therapies, ensuring that hospital programs for cell treatments, such as CAR T-cell therapy, follow rigorous protocols and measure outcomes. This group, the Foundation for the […]
View Blog