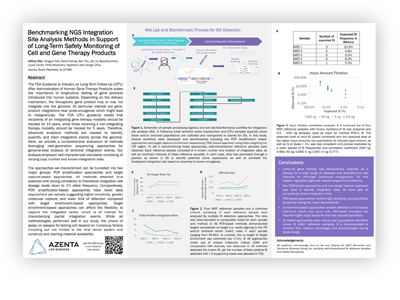
Benchmarking NGS Integration Site Analysis Methods in Support of Long-Term Safety Monitoring of Gene Therapy Products
The FDA Guidance to industry on Long Term Follow-Up (LTFU) after administration of human gene therapy products states the importance of longitudinal testing of gene products introduced into human subjects. Depending on the delivery mechanism, the therapeutic gene product may or may not integrate into the genome and recipients of an integrating gene therapy modality should be tracked for 15 years, while those receiving a non-integrating therapy modality should be tracked for 5 years. Here, we provide a comprehensive evaluation of methods leveraging next generation sequencing approaches for genome-wide analysis of lentiviral integration events. Our analysis employed well-characterized standards consisting of varying copy number and known integration sites.
Download the poster to better understand the capabilities and uses of different integration site analysis techniques for patient safety testing in cell and gene therapeutic development.